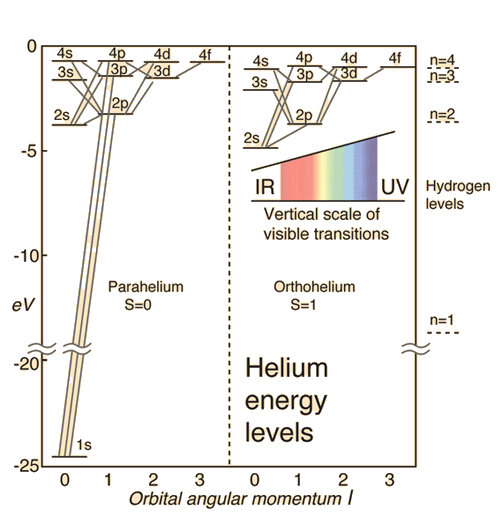
26. The ionisation energy of an electron in the ground state = 2 III =3 to n =1 (d) From n = 4 to n=3 of helium atom is 24.6 eV. The
Ionisation energy of He +is 19.6 × 10 18 J atom 1. The energy of the first stationary staten=1 of Li 2+ is:A. 8.82 × 10 17 J atom 1B. 4.41 × 10 16 J atom 1C. 4.41 × 10 17 J atom 1D. 2.2 × 10 15 J atom 1

The ionisation energy of hydrogen atom is 13.6 eV, the ionisation energy of helium atom would be (1988) (a) 13.6 eV (b) 27.2 eV (c) 6.8 eV (d) 54.4 eV
Helium Chemical Element First Ionization Energy Stock Vector (Royalty Free) 1220935573 | Shutterstock

With the help of Bohr \'s model , calculate the second ionisation energy of helium (energy requi... - YouTube

The first and second ionization potentials of helium atoms are 24.58 eV and 54.4 eV respectively... - YouTube
How to calculate the first ionization energy? Is it fixed? What is the difference between the first and second ionization energies? How do I calculate the second ionization energy - Quora

Defining first ionisation energy The first ionisation energy is the energy required to remove the most loosely held electron from one mole of gaseous atoms. - ppt download

![Kannada] The ionisation energy of an electron in the ground state of Kannada] The ionisation energy of an electron in the ground state of](https://static.doubtnut.com/ss/web-overlay-thumb/4038764.webp)