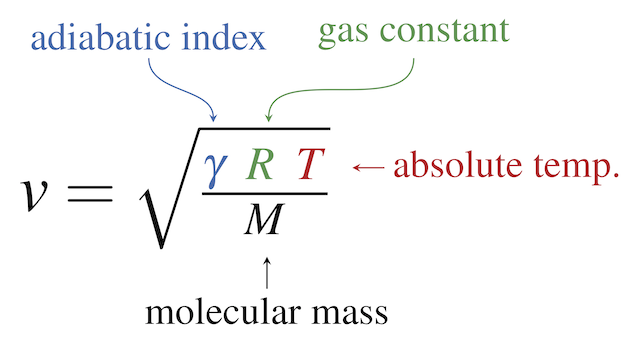
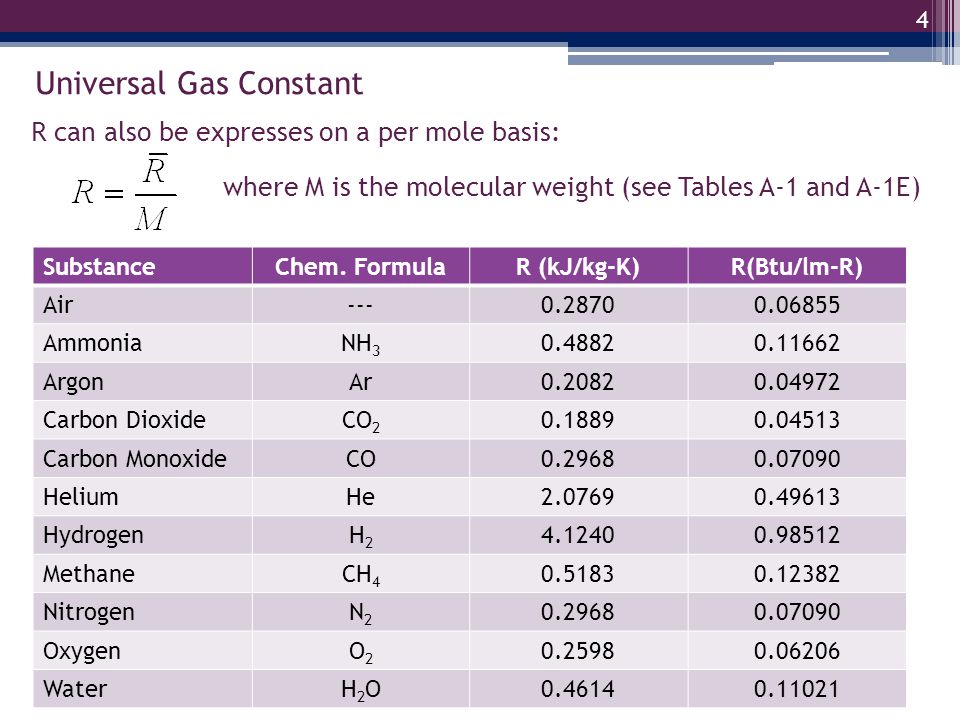
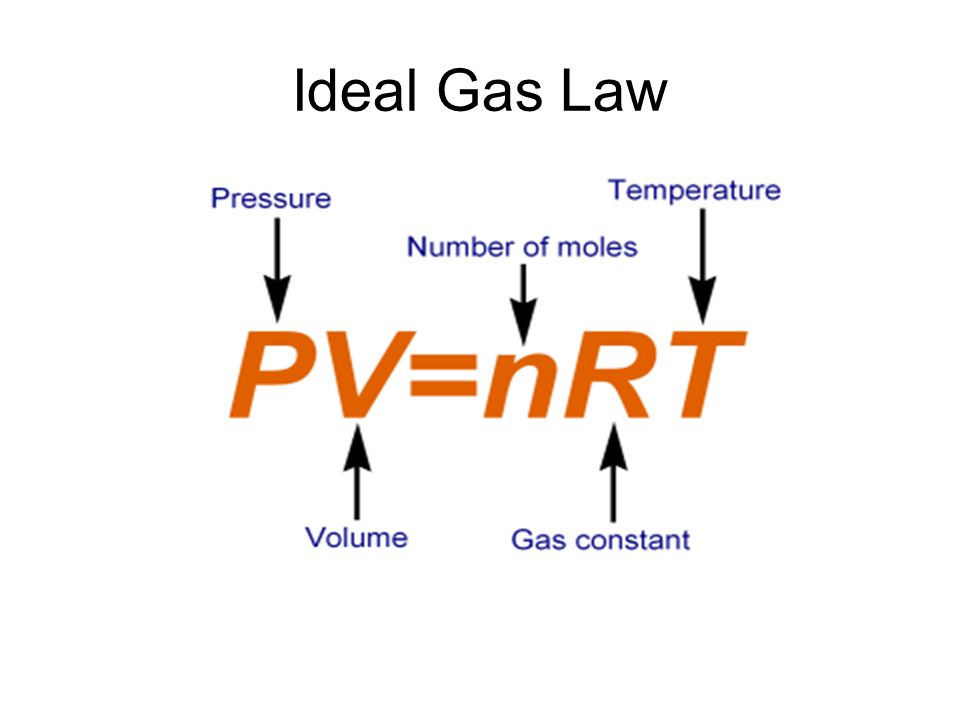
PDF) Determination of the Boltzmann constant k from the speed of sound in helium gas at the triple point of water

16 grams of helium gas (He) applies 380 mmHg pressure at a temperature of 0 °C. Assume ideal gas behavior. - brainly.com
![Two moles of Helium gas are to be taken over the cycle \\[{\\text{ABCDA}}\\]as shown (below) in the \\[{\\text{P - T}}\\]diagram. (Assume the gas to be ideal and R is a gas constant.).Now, Two moles of Helium gas are to be taken over the cycle \\[{\\text{ABCDA}}\\]as shown (below) in the \\[{\\text{P - T}}\\]diagram. (Assume the gas to be ideal and R is a gas constant.).Now,](https://www.vedantu.com/question-sets/0cb3c7f1-24ca-4773-848c-92578a8aa4e55598971945812544228.png)
Two moles of Helium gas are to be taken over the cycle \\[{\\text{ABCDA}}\\]as shown (below) in the \\[{\\text{P - T}}\\]diagram. (Assume the gas to be ideal and R is a gas constant.).Now,
What is the pressure (atm) of 3.5 moles of helium at -50 °C in a rigid container whose volume is 25.0 L? | Socratic

In a thermodynamic process helium gas obeys the law TP2/5=constantt. The heat given 'n' moles of He in order to raise the temperature from T to 2T is.

A balloon is filled with 1 mole of helium gas at 100 kPa of pressure and a temperature of 300 K. What is - brainly.com

Hot Selling Helium Balloon Gas 99.999% Helium Gas Cheap Price - China Helium Gas Constant, Helium Gas Cost | Made-in-China.com
Helium (He) gas is heated from 200 K to 400 K at constant pressure, the entropy change for 1 mole of the gas will be how to solve this problem?

The picture (not to scale) shows a pV diagram for 3.6 g of a helium gas (He) that undergoes the process 1 to 2 to 3 to 1. The ideal gas constant